Pharmaceutical Companies Partnering with IVD Manufacturers to Develop Companion Diagnostic Tests for their Therapeutic Drugs
Growing acceptance of companion diagnostics is a trend with the potential to greatly increase the value that clinical pathology laboratory testing delivers to physicians, patients, and payers. In 2010, it was increasingly common to see a pharmaceutical company announce an agreement with an in vitro diagnostics (IVD) manufacturer to develop a companion diagnostic test specifically for a therapeutic drug under development by that pharmaceutical company.
As most pathologists and clinical managers know, use of a companion diagnostic test is expected to add precision to the physician’s decision to prescribe therapeutic drugs. Thus, the increased number of public announcements during 2010 about companion diagnostic test development deals involving a pharmaceutical company and an IVD manufacturer indicates this trend is establishing solid roots.
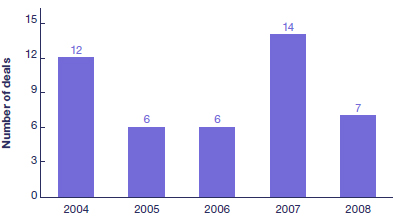
Companion Diagnostics Partnerships with Pharma in 2004-2008 (Sourced from PricewaterhouseCoopers "Diagnostics 2009 - Moving Towards Personalised Medicine. What Will You Do?")
Laboratory Test Development Deals Tracked by Dark Daily During 2010
Early in the year, Pfizer, Inc., (NYSE: PFE) and QIAGEN N.V., (NASDAQ: QGEN) agreed to develop a companion diagnostic test for Pfizer’s investigative drug PF-04948568 (CDX-110), which is an immunotherapy vaccine that treats glioblastoma multiforme (GBM). GBM involves brain tumors in adults and affects 25,000 people globally each year.
According to Pfizer’s press release, “PF-04948568 (CDX-110) is a peptide vaccine which targets the tumor-specific Epidermal Growth Factor Receptor variant III (EGFRvIII), a mutated form of the epidermal growth factor receptor that is only present in cancer cells and occurs in 25% to 40% of GBM tumors. The QIAGEN assay is designed to identify those patients whose tumors express the EGFRvIII mutation, allowing for the possibility of more targeted and personalized treatment.
“The EGFRvIII companion diagnostic will be developed and manufactured at QIAGEN’s Center of Excellence for Companion Diagnostics in Manchester, U.K.,” the press release continued, “The diagnostic will be a real-time PCR assay used to detect EGFRvIII RNA in tumor tissue. The assay is designed to offer a simple workflow, which supports its clinical utility in routine mutation testing.”
Similarly, just weeks later, Merck & Company, Inc., (NYSE: MRK) issued a press release announcing a research collaboration with Roche Holdings (PINK: RHHBY) to investigate ways to use Roche’s developmental microarray-based AmpliChip p53 test, which detects mutations in the tumor suppressor gene p53, to predict patients’ responses to specific therapeutic treatments.
Soon thereafter, GlaxoSmithKline PLC, (NYSE: GSK) disclosed that it was entering into an agreement with Abbot Laboratories (NYSE: ABT) to “develop a molecular diagnostic test intended for use as an aid in selecting patients who may benefit from a skin cancer treatment in development by GSK,” according to an Abbot press release.
Two Observations about Companion Diagnostics Development
Dark Daily subscribers and readers should focus on two aspects of the companion diagnostics trend. First, even as pharmaceutical companies bring drugs into clinical trials, they are now taking steps to simultaneously develop companion diagnostic tests. That way, when the clinical trials are completed, the pharma company has an appropriate companion diagnostic test to be used with its drug. It can then submit the therapeutic drug and companion diagnostic test to the Food & Drug Administration (FDA) to obtain market clearance for both products.
Secondly, there are examples where pharma companies approached IVD manufacturers specifically to develop a companion diagnostic test that would be performed on that IVD company’s FDA-cleared molecular testing instrument system. The pharma company benefits from this arrangement because the companion diagnostic would be an FDA-cleared medical laboratory test run on an FDA-cleared analyzer. For the pharma company, this would mean greater accuracy and reproducibility of results from the companion diagnostic test across all clinical laboratories and pathology groups using that companion diagnostic test kit.
The three companion diagnostic deals described above demonstrate that the pace of progress in the development and use of companion diagnostic tests is building momentum. Of course, it will be several more years before the pharmaceutical companies can complete clinical trials for the drugs associated with the companion diagnostic tests now under development. However, these joint development efforts between pharma companies and IVD manufacturers show how the need for companion diagnostics is likely to increase the role of the pharmaceutical industry in the pathology profession and in clinical laboratory testing going forward.
Related Information:
Roche Collaborates with Merck on Developmental Test for Cancer-Related Gene Mutation



