The ATC Chip identifies ovarian cancer cells floating in ascites and may be useful for diagnosing other types of malignancies that involve ascites, like pancreatic cancer
Pathologists will be interested to learn that researchers at Harvard Medical School and Massachusetts General Hospital are developing a “liquid biopsy” technology specifically to enable point-of-care monitoring of the progress of patients undergoing treatment for certain types of cancers.
The goal is to develop a method that community hospitals can use to monitor treatment of ovarian cancer patients without the need for expensive medical laboratory equipment, noted a report published by Biosciencetechnology.com. Researchers estimate that their ‘liquid biopsy’ technology could cost as little as $1 per test when eventually cleared for use in clinical settings.
The research team created a microchip device—called the ATC chip—that simplifies monitoring of a patient’s response to treatment for ovarian cancer and certain other malignancies. The device captures, isolates, and identifies tumor cells from ascites (ATCs).
Using Ascites as a Biomarker
Ascites are found in the fluid that frequently accumulates in the abdomen of patients with ovarian and abdominal cancers, noted Biosciencetechnology.com.
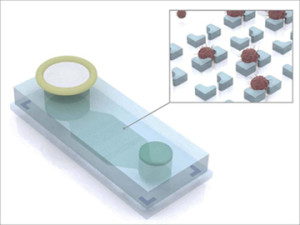
With the “liquid biopsy” device (pictured here) developed by researchers at Harvard University, ascites fluid from ovarian cancer patients flows through a filter into an inlet (left) where magnetically labeled benign cells are trapped. The remaining fluid passes through a microchip containing microwells that capture (insert) antibody-labeled tumor cells for collection, imaging and additional analyses. (Illustration copyright Hakho Lee and Jaehoon Chung, Massachusetts General Hospital Center for Systems Biology.)
Benefits of the Microchip-based Ovarian Cancer Monitoring Device
The ability to reliably track treatment response enables the patient’s clinician to determine if a particular cancer drug is effective or not. It also helps the clinician determine whether it may be appropriate to try another anticancer drug. Tumor recurrence begins before metastases become visible on imaging studies. That is why several options for non-invasive “liquid biopsies” are being investigated, noted the Biosciencetechnology.com article.
The device under development at Harvard does this at the point-of-care without the need for advanced clinical laboratory technology. “The ability to perform longitudinal testing of ascites in a point-of-care setting could significantly impact clinical trials, drug development, and clinical care,” noted authors of the paper published on the Proceedings of the National Academy of Sciences website (PNAS ). “Here, we developed a microfluidic chip platform to enrich ATCs from highly heterogeneous peritoneal fluid and then perform molecular analyses on these cells.”
‘Liquid Biopsy’ Device for Monitoring Ovarian Cancer Patients
The researchers noted that serial molecular analyses of tumor cells during treatment- and biopsy-driven clinical trials are emerging norms for many types of cancer. Yet surgical and image-guided biopsies are expensive and invasive. This is why alternative means for analyzing and tracking tumor cells are being sought.
“We were able to demonstrate that simply squirting small amounts of otherwise discarded ascites fluid into our device allowed us to quantify tumor cells and explore mechanistic marker of tumor progression without the need to process liters of ascites with advanced instrumentation not readily available at many community hospitals,” explained Cesar M. Castro, M.D., Instructor in Medicine in the Hematology/Oncology Department at Massachusetts General Hospital. He was quoted in the Biosciencetechnology.com report.
“Moreover, achieving point-of-care readouts of tumor cell markers from repeatedly collected ascites at different time points could allow for frequent monitoring of treatment response without having to wait for the next imaging scan,” added Castro.
Challenges in Using Ascites Fluid as a Cancer Biomarker
However, isolation of ascites tumor cells is challenging because they comprise just 1% of cells in ascites fluid. Additionally, investigators had to identify the ovarian cells and analyze their molecular properties to develop and test the four-protein-marker panel—named ATCDX—for accuracy. This was achieved with intensive laboratory work to compare ovarian cancer cells with benign cells and ascites samples from ovarian cancer patients and patients with noncancerous conditions, like cirrhosis, as well as with other types of cancers.
Before passing ascites fluid through the ATC chip, the sample is labeled with magnetic nanoparticles that bind to noncancerous inflammatory cells, noted the Biosciencetechnology.com article. The ATCs sample is passed through a filter into the three-inch-long ATC chip to screen out clumps of debris. It is then passed by a magnet that traps the magnetically labeled benign cells. A mixture of antibodies and the ATCDX proteins, which label the markers for imaging detection, also is added to the device. After the magnetic sorting, the remaining fluid passes through a microchip’s microwells, which capture antibody-labeled tumor cells for collection, imaging and additional analyses.
Using the ATC chip to analyze molecular properties of ascites tumor cells from 46 ovarian cancer patients, the Harvard researchers were able to determine which patients’ tumors had responded to treatment and which did not.
Device Exceeds Expectations and Will Cost Only About $1 Each
“This device far exceeded our expectations,” said Raph Weissleder, M.D., Ph.D., Thrall Family Professor of Radiology at Mass General and senior author of the PNAS paper. “Coupled with our diagnostic panel, we were able to clearly distinguish between tumor cells and the extensive cellular debris commonly found in ascites.
“The ATC chip and the set of protein markers we uncovered, which reliably identified ovarian cancer cells floating in ascites, provide a novel platform for extending ascites tumor cells analysis to settings where the expensive equipment and labor-intensive techniques that ascites tumor cells previously required would not be feasible,” concluded Weissleder.

Dr. Raph Weissleder, M.D., Ph.D., (pictured) Thrall Family Professor of Radiology at Boston’s Massachusetts General Hospital and Professor at Harvard Medical School, said the ATC Chip exceeded expectations, providing an novel, inexpensive platform for monitoring ovarian cancer patient response to treatment. (Photo copyright Harvard Stem Cell Institute.)
Large-scale production of the ATC chip is already being planned, according to the Biosciencetechnology.com report. Further studies are needed to confirm the results of this research. However, the device’s ease of use and low cost, estimated at $1 each, would make this a practical and valuable tool for both treatment and research of ovarian cancer and possibly other types of malignancies, like pancreatic cancer, that cause formation of ascites.
Biotechnology Developers Taking on Healthcare Reform
Efforts to develop “liquid biopsy” methods are being watched by the anatomic pathology profession. Both academic research centers and biotech companies have their eyes on developing more accurate diagnostic procedures that can be performed more cheaply than existing molecular diagnostic assays and genetic tests. They are also interested in using different types of specimens to generate an equal or better diagnostic result compared to existing diagnostic protocols for patients with ovarian cancer and other types of cancer.
—By Patricia Kirk
Related Information:
‘Liquid Biopsy’ Device Simplifies Cancer Monitoring
Ascites analysis by a microfluidic chip allows tumor-cell profiling
Clinical waste may prove valuable for monitoring treatment response in ovarian cancer




I have state III ovarian cancer and have recurrence twice since my intitial operation June 2012. The first one occurred in May 2013 after taking Carbo/Taxol for 18 weeks. I started on Advastin/Taxotol for 18 weeks. The cancer was gone according to a Cat Scan I had done on October 31, 2013. Then I have had the cancer to return in the last three weeks. I started having indgestion and some swelling in lower abdomen. When the Doctor did a Pet Scan the cancer was back and now just started Doxil with the first treatment last Tuesday. I am so excited to hear of the ACT Chip and what it can do. My CA125 is not a marker for me–each time it came back it was below 9. When will this chip be available for the masses or for a trial version?