Feb 21, 2018 | Instruments & Equipment, Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing, Management & Operations
It has been regularly demonstrated in recent decades that human breath contains elements that could be incorporated into clinical laboratory tests, so the decision to use this “breath biopsy” test in a therapeutic drug trial will be closely watched
When a major pharma company pays attention to a breath test, implications for clinical laboratories are often forthcoming. Such may be the case with GlaxoSmithKline (GSK). The global healthcare company has selected Owlstone Medical’s Breath Biopsy technology for use in its Phase II clinical trial of danirixin (DNX), a respiratory drug under development by GSK for treatment of chronic obstructive pulmonary disease (COPD), an Owlstone Medical news release announced.
Anatomic pathologists and medical laboratory leaders will be intrigued by GSK’s integration of breath-based specimens in a clinical trial of a respiratory drug. The partners in the trial aim to analyze breath samples to better understand the drug’s treatment effects and to discover personalized medicine (AKA, precision medicine) opportunities.
GSK (NYSE:GSK), headquartered in the UK but with a large presence in the US, researches and develops pharmaceutical medicines, vaccines, and other consumer health products.
Owlstone Medical, a diagnostic company, is developing a breathalyzer for disease and says it is on a mission to save 100,000 lives and $1.5 billion in healthcare costs. Dark Daily previously reported on Owlstone Medical’s Breath Biopsy platform. The Cambridge, England-based company has raised significant funding ($23.5 million) and already garnered credible cancer trial collaborators including the UK’s National Health Service (NHS).
Now, Owlstone Medical has brought its breath analysis technology to bear on chronic disease outside of cancer diagnostics development. A pharmaporum article called Owlstone’s Medical’s work with GSK an “additional boost of confidence” in the company’s technology, as well as a means for revenue.

Billy Boyle, co-founder and Chief Executive Officer, Owlstone Medical (above), shown with the company’s ReCIVA Breath Sampler device. This will be used by GSK in its Phase II respiratory disease clinical trial of danirixin to “capture VOC biomarkers in breath samples.” (Photo copyright: Business Weekly UK.)
GSK Studying Future Treatments for Respiratory Diseases
COPD affects about 700 million people worldwide, an increase of about 65% since 1990, GSK pointed out. In September 2017, GSK presented respiratory disease data and its pipeline medications at the European Respiratory Society in Milan, Italy. Included was information on danirixin (an oral CXCR2 antagonist), which is part of the company’s focus on COPD disease modification, according to a GSK news release.
“Each of our studies sets the bar for our future research and innovation,” noted Neil Barnes, MA Cantab, FRCP, FCCP(Hon), Vice President, Global Franchise Medical Head, GSK Respiratory, in the GSK press release.
Clinical Trial Aimed at Identifying the ‘Right’ Patients
With Owlstone Medical’s breathalyzer, GSK plans to explore how volatile organic compounds (VOCs) can help identify patients who will benefit most from the company’s medications, as well as evaluate Danirixin’s effects. A critical element of personalized medicine.
“It’s part of our efforts to identify the right patient for the right treatment,” said Ruth Tal-Singer, PhD, GSK’s Vice President of Medicine Development Leader and Senior Fellow, Respiratory Research and Development, in the Owlstone Medical news release.
VOCs in breath will be captured in a non-invasive way from patients who wear Owlstone Medical’s ReCIVA Breath Sampler, which, according to Owlstone Medical, has CE-mark clearance, a certification noting conformity with European health and safety standards. The VOCs breath samples will then be sent to Owlstone Medical’s lab for high-sensitivity analysis.
“Non-invasive Breath Biopsy can establish a role in precision medicine applications such as patient stratification and monitoring treatment response,” said Billy Boyle, Owlstone Medical’s co-Founder and Chief Executive Officer.
VOC Biomarkers in Respiratory Disease
VOC profiles can be characteristic of COPD as well as other respiratory diseases including asthma, tuberculosis, and cystic fibrosis, reported Science/Business.
According to Owlstone Medical’s Website, VOCs are gaseous molecules produced by the human body’s metabolism that are suitable for Breath Biopsy. Their research suggests that exhaled breath reflects molecular processes responsible for chronic inflammation. Thus, VOCs captured through Breath Biopsy offer insight into respiratory disease biomarkers.
Breath also includes VOCs that originate from circulation, which can provide information on a patient’s response to medications.
How the Breath Biopsy Platform Works
Owlstone Medical’s platform relies on its patented Field Asymmetric Ion Mobility Spectrometry (FAIMS) technology, which “has the ability to rapidly monitor a broad range of VOC biomarkers from breath, urine and other bodily fluids with high sensitivity and selectivity,” according to the company’s website. During the process:
- Gases are exchanged between circulating blood and inhaled fresh air in the lungs;
- VOC biomarkers pass from the circulation system into the lungs along with oxygen, carbon dioxide, and other gases;
- Exhaled breath contains exiting biomarkers.
It takes about a minute for blood to flow around the body. So, a breath sample during that time makes possible collection and analysis of VOC biomarkers from any part of the body touched by the circulatory system.
The medical analysis is enabled by software in the Owlstone Medical lab, Boyle told the Cambridge Independent.
“There’s an analogy with blood prints—you get the blood and can look for different diseases, and we’ve developed core hardware and technology to analyze the breath sample,” he said.
Another Breath Sample Device
The ReCIVA Breath Sampler is not the only breathalyzer focused on multiple diseases. Dark Daily reported on research conducted by Technion, Israel’s Institute of Technology, into a breath analyzer that can detect up to 17 cancers, and inflammatory and neurological diseases.
But Owlstone Medical stands out due, in part, to its noteworthy partners: the UK’s National Health Service, as well as the:
And now the company can add collaboration with GSK to its progress. Though some question the reliability of breath tests as biomarkers in the areas of sensitivity and specificity required for cancer diagnosis, Owlstone Medical appears to have the wherewithal to handle those hurdles. It is a diagnostics company that many pathologists and medical laboratory professionals may find worth watching.
—Donna Marie Pocius
Related Information:
Owlstone Medical’s Breath Biopsy Platform Integrated into GSK’s Phase II Respiratory Disease Clinical Trial
GSK Utilizes Owlstone Disease Breathalyser for Key Clinical Trials
GSK Presents Respiratory Data from Pipeline to Clinical Practice at ERS
GSK Boosts Medtech First Owlstone with Use of Breath Biopsy in Respiratory Trial
Glaxo to Stratify COPD Trial Using Breath Biopsy Device
Billy Boyle of Owlstone Medical on the Inspiration Behind His Mission to Save 100,000 From Dying of Cancer
Owlstone Medical and UK’s NHS Study Whether Breath Contains Useful Biomarkers
Breath Based Biomarker Detection: Informing Drug Development and Future Treatment Regimes
Clinical Laboratories Could Soon Diagnose 17 Diseases with a Single Breath Analyzer Test from Israel’s Institute of Technology
Feb 14, 2018 | Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing
Tight provider networks have some seniors dropping private plans after losing access to ‘preferred doctors and hospitals’ and experiencing issues with ‘access to care’
Medicare Advantage Plans continue to rise in popularity. That trend has implications for clinical laboratories and anatomic pathology groups because private insurers running Medicare Advantage plans tend to have narrow or exclusive lab networks.
Thus, as Medicare patients shift from Medicare Part B (which pays any provider a fee-for-service reimbursement) to a Medicare Advantage plan (with a narrow network), labs in that community lose access to that patient.
Now a recent government study of the Medicare Advantage program has interesting findings. For seniors in poor health, the private healthcare plans can prove costly if they lose access to specialized healthcare and the freedom to go to any doctor or hospital.
High Turnover Could Mean Poor Quality Plans
A 2017 report by the Government Accountability Office (GAO) found that beneficiaries in poor health are more likely to disenroll from Medicare Advantage Plans—a sign that the quality of plans with higher than normal turnover may be poor. The agency reviewed 126 Medicare Advantage plans and found that 35 of them had disproportionately high numbers of sicker people dropping out. Many seniors cited problems with “coverage of preferred doctors and hospitals” and “access to care.” The GAO is urging the Centers for Medicare and Medicaid Services (CMS) to review disenrollment data by health status and disenrollment reasons as part of the agency’s routine monitoring efforts.
“People who are sicker are much more likely to leave [Medicare Advantage plans] than people who are healthier,” James Cosgrove, Director of Healthcare at the GAO, told Kaiser Health News.
David Lipschutz, JD, Managing Attorney at the Center for Medicare Advocacy, called for tighter government oversight of Medicare Advantage plans.
“A Medicare Advantage plan sponsor does not have an evergreen right to participate in and profit from the Medicare program, particularly if it is providing poor care,” Lipschutz told Kaiser Health News.

David Lipschutz, JD (above), Managing Attorney for the Center for Medicare Advocacy, is calling for tighter oversight of Medicare Advantage Plans following a report by the Government Accountability Office (GAO) showing an exodus of sicker patients from some Medicare Advantage plans. (Photo copyright: Center for Medicare Advocacy.)
Threat to Regional Medical Laboratories by Narrow Networks
Dark Daily previously reported on how enrollment shifts from traditional Medicare to Medicare Advantage threaten the financial health of regional clinical labs, which typically lose access to Medicare Advantage beneficiaries. In 2017, one in three (33%) Medicare beneficiaries was enrolled in a private Medicare Advantage plan, reflecting 8% growth (1.4 million beneficiaries) between 2016 and 2017, according to a Kaiser Family Foundation (KFF) report.
Medicare Advantage’s private health plans are attractive to many seniors because of lower cost sharing and expanded benefits, such as hearing aid and eyeglass coverage and fitness club memberships. The tradeoff, however, requires forfeiting access to Medicare Part A (hospital insurance) and Part B (medical insurance) and accepting a narrower network of providers and hospitals.

An analysis by the Kaiser Family Foundation shows that more than three in 10 (35%) of Medicare Advantage enrollees in 2015 were in narrow-network plans. On average, Medicare Advantage networks included less than half (46%) of physicians in a county. The size and composition of Medicare Advantage Provider networks greatly impacts smaller clinical laboratories and anatomic pathology groups, which often are excluded from narrow-network plans. (Image copyright: KFF.)
Ron Brandwein, Health Insurance Information, Counseling and Assistance Program Coordinator at Lifespan of Greater Rochester, N.Y., believes consumers need to understand the limitations of Medicare Advantage plans.
“It’s very competitive, very dog eat dog,” he told the Democrat and Chronicle, adding that, once a person signs up with a Medicare Advantage plan, all their dollars for care are sent to that plan. “If they wind up going to a doctor or hospital that doesn’t accept it, they can’t fall back on Medicare because Medicare won’t pay their bills anymore because they’ve given their dollars to their chosen Advantage plan,” he said.
The 2017 KFF report “Medicare Advantage: How Robust Are Plans’ Physician Networks,” found:
- One in three Medicare Advantage enrollees in 2015 were in a plan with a narrow physician network (less than 30% of physicians in the county);
- 43% were in medium-sized networks (30% to 69% of physicians in the county); and,
- Just 22% were in broad plans that included 70% or more of physicians in the county.
“Insurers may create narrow networks for a variety of reasons, such as to have greater control over the costs and quality of care provided to enrollees in the plan,” KFF reported. “The size and composition of Medicare Advantage provider networks is likely to be particularly important to enrollees when they have an unforeseen medical event or serious illness. However, accessing the information may not be easy for users, and comparing networks could be especially challenging. Beneficiaries could unwittingly face significant costs if they accidentally go out-of-network.”
But Kristine Grow, Senior Vice President, Communications, at America’s Health Insurance Plans (AHIP), contends most consumers are satisfied with their Medicare Advantage plans, as evidenced by the growth in Medicare Advantage enrollment. She told Kaiser Health News that patients in the GAO study mostly switched from one health plan to another to take advantage of a better deal or more inclusive coverage.
“We have to remember these are plans working hard to deliver the best care they can,” Grow said. Insurers compete vigorously for business and “want to keep members for the longer term,” she added.
Smaller Clinical Laboratories at Greatest Risk
The implications for anatomic pathology groups and medical laboratories is clear. As Dark Daily has reported, increasing reliance by insurers on narrow networks to stem raising costs limits the number of physicians ordering medical testing, reducing lab revenues and threatening the entire pathology industry—especially smaller clinical laboratories. And, since Medicare patients now represent more than 50% of all patients in the healthcare system, the impact of that aging population’s behavior increases each year.
—Andrea Downing Peck
Related Information:
As Seniors Get Sicker, they’re More Likely to Drop Medicare Advantage Plans
Medicare Advantage 2018 Data: First Look
Medicare Choices More Complicated for Seniors Who Use Rochester Regional Health
Medicare Advantage: How Robust Are Plans’ Physician Networks?
Medicare Advantage Plans in 2017: Short-term Outlook is Stable
Medicare Advantage: CMS should Use Data on Disenrollment and Beneficiary Health Status to Strengthen Oversight
Sustained Growth in Medicare Advantage Plans Threatens Financial Health of Smaller Pathology Groups and Local Medical Laboratories
Kaiser Family Foundation Study Predicts Big Increases in Obamacare Premiums for 2017; However, Narrow Networks Often Exclude Clinical Laboratories and Other Providers
McKinsey Study Confirms Trend Toward Narrow Healthcare Networks on Health Insurance Exchanges; Smaller Clinical Laboratories and Pathology Groups Often Excluded
Narrow Networks Mean Shrinking Opportunity for Pathology and Clinical Medical Laboratories
Feb 12, 2018 | Digital Pathology, Instruments & Equipment, Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing
Doctors’ advocacy organization praises potential of ‘My Health Record’ but voices concerns about functionality, interoperability, and added burden placed on providers
Australia’s goal of implementing a nationwide electronic health record (EHR) system received a major boost when the country’s largest pathology laboratories signed agreements with the Australian Digital Health Agency (ADHA) to join the project. But the My Health Record system has yet to fully win over providers as the Australian Medical Association (AMA) raises concerns over functionality, interoperability, and the added burden placed on healthcare providers.
ADHA Chief Executive Tim Kelsey praised the addition of pathology and diagnostic organizations to the My Health Record platform. In Australia, pathology laboratory is the term to describe what are called clinical laboratories in the United States.
“The largest diagnostic organizations in Australia have now agreed to share their test reports with Australian consumers,” Kelsey said in an ADHA news release. “We are working to deliver a My Health Record for all Australians next year, unless they choose not to have one. Health consumers will benefit from this significant commitment by the pathology industry and their software partners.”

Australian Digital Health Agency CEO Tim Kelsey says his agency in 2018 will be creating a universal electronic health record for all the country’s 24.8 million citizens, though patients will have the option to opt out of the My Health Record project. He called the “significant commitment” of pathology labs to the project a major step forward. (Photo copyright: ADHA.)
In May 2017, Sonic Healthcare, Australia’s largest pathology provider, became the first private pathology company to join the My Health Record initiative. That news was followed by agreements between the ADHA and pathology companies Primary Health Care, Australian Clinical Labs, and seven other software vendors and pathology laboratories, including:
The ADHA also finalized service agreements with additional software companies that will enable diagnostic imaging providers to link up to My Health Record by the end of 2018.
AMA Says My Health Record Lacks Functionality and Critical Features
In 2012, Australia announced the roll out of the Personally Controlled Electronic Health Record, the original initiative to create a citizen-controlled secure online summary of health information, which later was renamed My Health Record. According to The Australian, more than 5.3 million Australians are now using My Health Record, a 500% increase in the number of shared health summaries uploaded in 2016-2017 and a 200% rise in interoperability with private hospitals.
Royal College of Pathologists of Australasia President Bruce Latham, MBBS, welcomed the announcement of the increased functionality for My Health Record.
“The Australian pathology sector has been working in support of the national eHealth agenda for a number of years,” Latham stated in the ADHA news release. “Work is now progressing to connect both public and private labs to the My Health Record, and patients nationally will start to see their pathology reports in their My Health Record.”
Developers and program administrators of My Health Record predict it will generate savings of AU$123 million from:
- Reduction in adverse drug events;
- Fewer duplicated diagnostics tests; and,
- Cost savings by 2020-2021.
However, the AMA, Australia’s doctors’ advocacy group, outlined its concerns about My Health Record in a Pre-Budget Submission to the Australian federal government. While praising the project’s potential to “not only save money, but save lives,” the AMA argued the national repository of healthcare information needs improved features and functionality to meet its potential.
“… more work is required,” the AMA wrote. “The return on investment will hinge in the short term on ease of use for medical practitioners who upload the clinical data. Interoperability with the multiple software packages used across the medical profession and broader health sector must be seamless.
“Problems uploading specialists’ letters, poor search functionality, time-consuming adaptations to existing medical practitioner work practices, or inappropriate workarounds will erode clinical utility and deter doctor use—and, more importantly, take time away from focusing on the patient,” the AMA concluded.
Automatic Enrollment Concerns AMA
My Health Record began as a self-register model, but as the program goes nationwide in 2018, it will do so using an “opt-out” model. This means citizens will be enrolled automatically unless they ask to be removed from the program. According to the ADHA, the automatic creation of My Health Record for all Australians will begin in mid-2018. The government’s goal is to provide access to My Health Record to all 24.6 million Australians by June 30, 2018.
The federal government’s switch to an opt-out system for My Health Record drew concerns from the AMA.
“Doctors do not have time to talk their patients through the My Health Record arrangements for opt-out, privacy, [or] setting access controls in standing consent for health providers to upload health information. This is the work of the government. Doctors must be allowed to focus on what they do best—caring for patients,” the AMA stressed.
Clinical Laboratories Have Stake in Outcome
According to Healthcare IT News Australia, the Australian government has spent AU$2 billion ($1.53 billion USD) so far developing what could become a white elephant if general practitioners and hospital groups don’t see a clinical benefit in its use.
If Australia is successful in creating a fully-functioning and widely-used national repository for health information, it will be among the first countries to do so. In 2002, the United Kingdom (UK) kicked off a nearly decade-long effort to create a national EHR system for the UK’s single-payer tax-supported health system. Ultimately, the government pulled the plug on the initiative after spending 12.7 billion pounds ($17 billion USD) trying to complete the project.
That result, and lessons learned from Australia’s experience, should inform American healthcare policy makers. It remains a daunting effort to implement a single electronic patient health records system. Of course, pathologists and clinical laboratory administrators have an interest in this issue, since medical laboratory test data represents the largest proportion of an individual patient’s permanent health record.
—Andrea Downing Peck
Related Information:
Australia’s Largest Pathology Labs Sign Up to My Health Record
Private Pathology Reports to Go Live in My Health Record
E-health Revolution Gather Pace as more Services Pledge their Backing
Australian Medical Association Pre-Budget Submission 2018-19
National Expansion of My Health Record in 2018 Confirmed in Budget Announcement
GPS and Hospitals Claim My Health Record Not Fit for Purpose as Alarming Low Usage Figures Are Released
Feb 9, 2018 | Laboratory Instruments & Laboratory Equipment, Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing
Genalyte’s cloud-based Maverick Detection System could potentially change how and when doctors order blood draws, altering long-standing clinical laboratory workflows
Anatomic pathologists and medical laboratory leaders may be aware of efforts in the in vitro diagnostics (IVD) industry to perform clinical laboratory tests with smaller quantities of blood. The most high-profile company to try and fail is Theranos, which both Dark Daily and its sister print publication The Dark Report reported on as events unfolded.
So far, though, continued efforts to dramatically reduce the amount of blood needed for most typical medical laboratory tests have come up dry. But this has not stopped innovative companies from trying to do so.
One such company is San Diego-based Genalyte. The biomedical diagnostics developer has announced it is readying its new Maverick Detection System (Maverick), which, according to the company’s website, “completes a comprehensive battery of blood tests in the physician’s office with results in 15 minutes.”
According to a news release, “Genalyte is laying the groundwork to move the business of biomedical diagnostics online, with the idea of creating an integrated delivery service for test results that can be generated from a drop of blood.” If successful, Maverick may be poised to disrupt the phlebotomy and clinical laboratory industries in a big way.
Fifteen Minutes from Fingerprick to Clinical Lab Test Results
Maverick, according to its developers, “[will] send digital samples to the cloud for quality review before releasing to the physician and patient. Our central lab handles tests that cannot be completed onsite.
“At the core of our cloud-based, diagnostic laboratory offering is revolutionary technology that uses silicon photonic biosensors to perform multiple tests off a single drop of whole blood in 15 minutes,” notes Genalyte’s website.
In a MedCity News article, Cary Gunn, Genalyte’s founder and CEO, said, “There will always be a need for esoteric testing that needs to be referred to a laboratory. But for the vast majority of routine testing, there’s no reason why that can’t be done in the doctor’s office.”
How Maverick Completes Medical Laboratory Tests in Doctor’s Offices
According to Genalyte’s website, “The Maverick Detection System performs real-time detection of macromolecules in crude samples using biologically functionalized silicon photonic biosensors lithographically printed on disposable silicon chips.”
About the width of a pencil erasure, Maverick biosensor chips “are individually functionalized with unique probe molecules and are individually interrogated, making highly multiplexed analysis possible. As a sample flows over the chip, the probes on the sensors bind with their corresponding ligands. This binding results in a localized change in refractive index on the sensor surface; this change is directly proportional to analyte concentration.”
“The silicon chip itself is watching the chemical reactions take place. Anytime two molecules bind, we can see that happen. So, the technology is capable of almost an infinite number of tests,” Gunn explained in the MedCity News article.
According to the developer, test results are available “in 10-30 minutes depending on the type of assay performed.”

Cary Gunn, PhD, Genalyte’s Founder and Chief Executive Officer, said in a news release that the San Diego-based biomedical diagnostics company wants “to put a rapid and powerful suite of diagnostic tests in every physician’s office.” (Photo copyright: Genalyte.)
Pilot Studies Show Test Feasibility in Doctor’s Offices
The company also announced completion of two pilot studies of the platform’s effectiveness in performing anti-nuclear antibody (ANA) testing. The purpose of study “one” was to “evaluate the feasibility of using this novel instrument to perform ANA 8 tests in the clinic and to compare those results to the same sample tested in Genalyte’s CLIA registered laboratory.” Study “two” focused on “Detection of anti-nuclear antibodies for the diagnosis of connective tissue diseases (CTD).”
The ANA test is often ordered by physicians for diagnosis of CTDs, including:
• Rheumatoid arthritis;
• Systemic lupus erythematosus;
• Raynaud syndrome; and,
• Systemic scleroderma, according to an article in Rheumatoid Arthritis News.
“We are starting with rheumatology, but I call that our entry point,” Gunn told MedCity News. “Our goal is to decentralize the vast majority of diagnostic testing to be near the patient and near the physician.”
The two studies together involved about 750 patients, who were tested by Genaltye’s Maverick system over four months. Results of their blood tests, via fingerprick in the doctor’s office, were compared to traditional medical laboratory procedures and patient diagnoses.

According to the Genalyte video above, “The Maverick Detection System … directly detects the binding of proteins or antibodies to the sensor in real-time and results are analyzed simultaneously with the accompanying Genalyte software. Almost all of the most time consuming and expensive parts of assay development and sample testing are reduced or eliminated.” Click on the image to view the video. (Caption and video copyright: Genalyte.)
According to the news release and the published clinical abstracts, the researchers concluded that:
• Positive and negative results on whole blood tested on the Maverick system highly correlated with serum tested on previously approved devices;
• Multiplex ANA testing on whole blood in physician offices is feasible;
• Venous draw and fingerstick blood samples highly correlated; and
• Maverick has the propensity to improve patients wait times for diagnosis and to enhance their testing convenience.
“There is extremely high correlation for absolute value between venous blood and fingerstick blood, and between positive and negative results seen with whole blood on the Maverick and serum on the FIDIS Connective 10,” noted study “one” researchers.
“I’m impressed,” Patricia Jones, PhD, former President of the American Association for Clinical Chemistry (AACC), told Bloomberg News. “The game-changing part of this would be being able to do testing and potentially make a diagnosis immediately, instead of having to send out lab tests, wait several days, and then call the patient,” she added.
Can One Drop Do It All? Some Researchers Advise Caution
The controversy surrounding point-of-care fingerprick capillary blood draws performed on in-office automated blood analyzers, versus clinical laboratory venous draws performed on high-volume laboratory systems, is not new. Dark Daily has reported on several blood test studies in the past.
One such study involved bioengineers at Rice University. It concluded that fingerpricked capillary blood may not be accurate or reliable enough for clinical decision-making.
Their study acknowledged the value of such capillary blood testing in remote areas. But it also urged caution about use of measurements from a single drop of fingerprick blood.
“Using both a hematology analyzer and POC hemoglobinometer, we found the variability of blood component measures to be greater for successive drops of fingerprick blood than for multiple drops of venous blood,” the researchers wrote in The American Journal of Clinical Pathology (AJCP).
Research will no doubt continue until a viable, accurate, and affordable blood analyzer system that conducts dozens of clinical laboratory tests based on a few drops of blood comes to market. It’s basically inevitable in today’s world where computers can be built from molecules and miniature medical laboratories can be placed in chips, skin patches, and needles.
Pathologists and clinical laboratory leaders would be well advised to monitor the development of these various new diagnostic technologies. For most of the past decade, there has been a steady parade of companies and research teams announcing new discoveries that could revolutionize clinical diagnostics as performed today. However, few disruptive clinical laboratory tests or analyzers based on these technologies have made it into the clinical marketplace.
—Donna Marie Pocius
Related Information:
Can Genalyte Achieve What Theranos Touted?
Genalyte Takes Aim at Lab Testing Giants with Cloud-Based Service
Genalyte Raises $36 Million From Khosla for its One-Drop Blood Test
AACC President Calls Genalyte’s Blood Diagnostic Tests ‘Game-Changing’
Drop-to-Drop Variation in the Cellular Components of Fingerprick Blood: Implications for Point-of-Care Diagnostic Development
Genalyte Diagnostic Tool Shows Potential to Improve Turnaround Time in RA, Other Conditions
Application of a Novel Anti-Nuclear Antibody Multiplex Test Using Finger Stick and Venous Whole Blood in a Rheumatology Clinic—Demonstration of Feasibility
Rice University Researchers Publish Study About Variation in Drop-to-Drop Samples of Capillary Blood Collected by Fingerprick and Used for Clinical Laboratory Testing
After AACC Presentation, Elizabeth Holmes and Theranos Failed to Convince Clinical Laboratory Scientists and the News Media about Quality of Its Technology
Score for Theranos After AACC: Fail
Feb 7, 2018 | Digital Pathology, Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing, Management & Operations
Federal agency receives input on eight focus areas as it looks for ways to enable providers ‘to design and offer new approaches to delivering care’
Medical laboratories and anatomic pathology groups preparing for the transition from fee-for-service healthcare will want to keep a close eye on the Centers for Medicare and Medicaid Services (CMS). The federal agency’s administrator plans to set a “new direction” for CMS as it shifts to value-based reimbursement models for Medicare services that could impact clinical laboratory revenues.
In an informal Request for Information (RFI), the Center for Medicare and Medicaid Innovation (CMMI) sought feedback on a “new direction to promote patient-centered care and test market-driven reforms that empower beneficiaries as consumers, provide price transparency, increase choices and competition to drive quality, reduce costs, and improve outcomes.”
CMS to ‘Move Away’ from Engineering Healthcare ‘From Afar’
The agency requested input on eight focus areas:
1. Increased participation in Advanced Alternative Payment Models (APMs);
2. Consumer-directed care and market-based innovation models;
3. Physician specialty models;
4. Prescription drug models;
5. Medicare Advantage innovation models;
6. State-based and local innovation;
7. Mental and behavioral health models; and,
8. Program integrity.
Comments from healthcare providers, clinicians, states, payers, and stakeholders were accepted through November 20, 2017.
In a Wall Street Journal (WSJ) op-ed, CMS Administrator Seema Verma explained the agency’s process moving forward. “We will move away from the assumption that Washington can engineer a more efficient healthcare system from afar—that we should specify the processes healthcare providers are required to follow,” she wrote.

CMS Administrator Seema Verma (above) plans to lead the Center for Medicare and Medicaid Innovation “in a new direction” and may be signaling a willingness to give providers more flexibility with value-based care payment models for Medicare services. (Photo copyright: Healthcare Dive.)
The RFI states the new model design will follow six guiding principles:
1. Choice and competition in the market;
2. Provider choice and incentives;
3. Patient-centered care;
4. Benefit design and price transparency;
5. Transparent model design and evaluation; and,
6. Small scale testing.
Providers Need Freedom to Design New Approaches to Healthcare
Verma said CMS plans to review all Innovation Center models to determine “what is working and should continue, and what isn’t and shouldn’t.” She voiced concern that the complexity of some of the current models may have encouraged consolidation in the healthcare system, resulting in fewer choices for patients.
“We must shift away from a fee-for-service system that reimburses only on volume and move toward a system that holds providers accountable for outcomes and allows them to innovate,” Verma wrote in the WSJ op-ed. “Providers need the freedom to design and offer new approaches to delivering care. Our goal is to increase flexibility by providing more waivers from current requirements.”
Actual Progress of Value-based Healthcare ‘Herky-Jerky’
In its reporting on the recent CMS announcements, Healthcare DIVE suggested that the U.S. Department of Health and Human Services (HHS) “is looking to make some potentially major changes” in value-based payment models.
However, Neil Smiley, CEO of Loopback Analytics, which assists healthcare organizations with managing outcome-based care, believes the transition to value-based care may face stiffer headwinds under the new administration. He points to an August CMS proposal that canceled some mandatory bundled payment programs and scaled back others as an indication that healthcare transformation could be slowing.
“The pace at which CMS committed to rolling out value-based care is fundamentally different from the pace we’re currently seeing,” he told Health IT. “The progress toward value-based care, instead of this steady momentum they expected, is more of a herky-jerky fashion.”
Modify, Don’t Abandon Existing Payment Models, suggests HCTTF
The Health Care Transformation Task Force (HCTTF), a 42-member industry consortium, was among the stakeholders who responded to CMS’ RFI. In a 22-page letter, the task force reiterated its support for the healthcare system’s transformation to value-based payment and care delivery, while outlining areas for improvements. The group urged CMS to continue to develop new models while modifying, rather than abandoning, existing models that show promise and need time to achieve a lasting return.
“We would like CMS to continue support for promising models while balancing the current portfolio with new, innovative payment models,” Clare Wrobel, Director of Payment Reform Models at HCTTF, told Home Health Care News. “[But] it would be a mistake to discard current models that providers have already invested in and are showing real promise.”
Smiley, meanwhile, suggests clinical laboratory managers, pathologists, and other healthcare providers keep watch as healthcare transformation continues to evolve.
“The fee-for-service model, love it or hate it, is not dying. The organism has adapted,” he told Health IT. “For those that were aggressive early adopters of value-based care and really believed what they were hearing, and have gone fully after value-based care, some of them may feel a little exposed. If they go too hard too fast, they may suffer economically if they misjudge the pace at which this moves.”
—Andrea Downing Peck
Related Information:
Centers for Medicare and Medicaid Services: Innovation Center New Direction
Medicare and Medicaid Need Innovation
CMS Seeks ‘New Direction’ for Innovation Center
Comprehensive Care for Joint Replacement Payment Model
Task Force Calls on CMS to Encourage Alternative Payment Models
CMS Request for Information: Innovation Center New Direction
Task Force Urges CMS to Preserve Value Based Payment Models
Feb 2, 2018 | Instruments & Equipment, Laboratory Instruments & Laboratory Equipment, Laboratory Management and Operations, Laboratory News, Laboratory Operations, Laboratory Pathology, Laboratory Testing, Management & Operations
Research goal was to isolate circulating tumor cells in venipuncture samples with improved purity compared to standard spiral chips
Many research teams are pursuing the goal of creating assays that detect circulating tumor cells (CTCs) that would allow earlier and more accurate diagnosis of cancer. Now comes news of a unique technology developed at the University of Michigan (U-M) Ann Arbor that showed promised in an early study.
The method of using CTCs to diagnose cancer in patients, while further analyzing specific characteristics of a given cancer case, shows promise as an innovative tool for clinical laboratories and oncologists. However, current approaches face challenges when it comes to proving accuracy and establishing thresholds that might indicate the need for further action.
Researchers at U-M believe they may have solved that problem. They created “Labyrinth,” a “label-free microfluidic device” that condenses 637mm of channels—including 11 loops and 56 corners—onto a 500μm-wide chip that uses inertia and Dean flow to separate white blood cells and CTCs from venipuncture samples at rates as high as 2.5ml per minute. These results improve upon the traditional spiral chip design.
Publishing their findings in Cell Systems, first author of the study Eric Lin, PhD, noted, “With the recent advances in tools for genomic characterization, it is more compelling than ever to look at the tumor heterogeneity to understand tumor progression and resistance to therapies. The Labyrinth device enabled high yields of CTCs without the bias induced by antibody-based selection, allowing the identification of true biological tumor heterogeneity.”
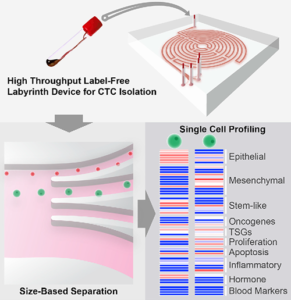
The graphic above, taken from the University of Michigan study, demonstrates the “High-throughput and label-free Labyrinth device that enables single CTC isolation and gene expression characterization.” According to the researchers, “Labyrinth offers a cell-surface marker-independent single-cell isolation platform to study heterogeneous CTC subpopulations.” The U-M study shows promise in creating tools for oncologist and clinical laboratory cancer treatment. (Image copyright: University of Michigan/Cell Systems.)
Challenges in the Isolation of CTCs
The Labyrinth chip is not the first device to assist in isolating CTCs. The U-M study notes that while immune-affinity capture is a validated approach to prognosis, therapeutic monitoring and molecular diagnostics, it does not work with all cancer cases. The researchers also note the method creates challenges in single-cell analysis later.
Existing label-free methods of isolation, such as deterministic lateral displacement, microfluidic flow fractionation, and acoustic-based separation, avoid these concerns but face issues of their own. The researchers noted, “Issues encountered with these approaches include pore clogging, high-pressure drop, pre-fixation to prevent CTC loss, low throughput, and excessive non-specific cell retention.”
The researchers further clarified that a major factor separating the Labyrinth chip from other methods is the ability to identify CTC subpopulations without the need for manual selection based on positive or negative protein expression. Thus, improving the ability to conduct further single-cell analysis from the results. Testing of the Labyrinth chip involved a variety of cancer cell lines, including:
· Human breast (MCF-7);
· Pancreatic (PANC-1);
· Prostate (PC-3); and,
· Lung (H1650).
And while standard spiral chips are already a common method for conducting size-based sorting, the purity of results is less than ideal with thousands of other cells remaining in the sample.
The researchers reported that the Labyrinth chip recovered 91.5% (plus or minus 0.9%) of cancer cells and removed 91.4% (plus or minus 3.3%) of white blood cells in a spiked buffer test.
“Bigger cells, like most cancer cells, focus pretty fast due to the curvature. But the smaller the cell is, the longer it takes to get focused,” Sunitha Nagrath, PhD, Associate Professor of Chemical Engineering and a lead developer of the Labyrinth chip, stated in a U-M news release. “The corners produce a mixing action that makes the smaller white blood cells come close to the equilibrium position much faster.”
Labyrinth also supports a series configuration of multiple chips. While testing two chips in series, researchers noted “a two-log improvement in tumor cell enrichment over the single Labyrinth.” They claim this is a higher purity than other label-free methods they studied, while adding only five minutes to processing times.

Sunitha Nagrath, PhD (above), is an Associate Professor of Chemical Engineering at the University of Michigan, and one of the lead developers of the Labyrinth chip. “You cannot put a box around these cells,” she noted in the U-M news release. “The markers for them are so complex, there is no one marker we could target for all these stages.” (Photo copyright: University of Michigan.)
Current Testing Using the Labyrinth Chip
The chip is already in use in a clinical trial for an aggressive form of breast cancer by Max Wicha, MD, Madeline and Sidney Forbes Professor of Oncology, Founding Director Emeritus, University of Michigan Comprehensive Cancer Center, and co-author of the Cell Systems study, who lead the study along with Nagrath.
The trial involves the attempted activation of adult system cells by blocking the signaling molecule interleukin-6. Wicha suspects the molecule enables cancer stem cells as well. “We think that this may be a way to monitor patients in clinical trials,” he said in the U-M news release. “Rather than just counting the cells, by capturing them, we can perform molecular analysis [to] know what we can target with treatments.”
The news release further highlights how this chip is specifically suited to such a task. As cancer stem cells transition from stem-like cells to more ordinary cell types, their gene expression shifts as well. This creates an issue when using conventional cell targeting. Nagrath notes this concern, stating, “The markers for [cancer stem cells] are so complex, there is no one marker we could target for all these stages.”
The Labyrinth chip shows potential for overcoming one of the biggest hurdles to leveraging CTCs to diagnose cancers and develop personalized therapies. Currently, the chip can output to Fluidigm, DEPArray by Silicon Biosystems, and RainDance Technologies’ RainDrop Digital PCR System.
The U-M researchers hope that future research will yield additional applications and compatible systems to further improve the ability for medical laboratories to use CTCs in the early detection and monitoring of cancer cases.
—Jon Stone
Related Information:
‘Labyrinth’ Chip Could Help Monitor Aggressive Cancer Stem Cells
High-throughput Microfluidic Labyrinth for the Label-free Isolation of Circulating Tumor Cells
Novel Labyrinth Chip Monitors Cancer Stem Cells in Clinical Trial
‘Labyrinth’ Device Sorts Cancer Cells from Healthy Blood
This Awesome Blood Labyrinth Is the Newest Method for Catching Cancer Cells
Labyrinth Chip Has the Potential to Lead to Customized Cancer Treatments












